Ovarian endometriosis occurs in 20-40% of patients with endometriosis (1). The most significant ovarian involvement is an endometrioma. Endometriomas occur when surface ovarian endometriosis lesions start to push into or invaginate into the ovary and then close over the top, forming a cystic mass lined by endometriosis tissue. Endometriomas are also called “chocolate” cysts. The presence of an endometrioma has significant implications as to the extent of endometriosis and to the potential impact on ovarian function and fertility.
As I have proctored or taught other surgeons, a statement that always worried me the most was to hear a surgeon say, “This surgery is just a quick endometrioma.” This allowed me to know that the surgeon did not have a full understanding of endometriosis as a disease and was woefully unprepared for the surgery at hand. In fact, the presence of an endometrioma in a patient with pain has a markedly increased risk of deeply infiltrating endometriosis elsewhere (2). Rarely is it “just an endometrioma.” It carries an increased risk of deep endometriosis in the uterosacral ligaments, intestine and vaginal wall.
There is much discussion about endometriomas and infertility. This discussion revolves around preserving fertility while attempting to “do no harm.” The main focus on fertility with respect to endometriomas is the impact they have on ovarian function and the ability to achieve a successful pregnancy. The presence of an endometrioma can have a negative impact on ovarian reserve or the number of ovarian follicles that contribute to the hormone cycle that allows ovulation (3). There are three main clinical markers used as outcomes for management of endometriomas: Anti-Mullerian Hormone (AMH), Antral Follicle Count (ANF) and Successful Pregnancy Rates (this is ultimately the most important outcome measure). Management of an endometrioma should be individualized to a patient’s goals and expectations. The discussion should include current and future goals related to pain, relationships and fertility. It is imperative that the patient also understands that prolonged observation with or without pain relief can allow the endometriosis to progress and negatively impact fertility further.
For some patients where pain is minimal and fertility is the ultimate goal, artificial reproductive technology can meet the goals and expectations. For others in whom fertility is not an immediate concern but a future one, or those with a significant pain component, hormone control of symptoms or surgical management may be better. To be clear, the only role of hormones in endometriomas is to try to help with symptoms. Hormones will not make endometriomas disappear, resolve, or “go away.”
Surgical management of endometriomas follows two lines of thought. These are actually similar to the approaches taken to surgical management of endometriosis in general, and we find similar results. One approach to surgical management is to drain the cyst with or without cauterizing or fulgurating the cyst wall. The other approach is called “cystectomy” or removal of the wall of the cyst.
Simple drainage of an endometrioma releases the non-active fluid, but leaves the actual endometriosis tissue in place, untreated. Some surgeons will attempt to burn or cauterize the cyst wall with hopes that it will destroy the active tissue of the endometrioma. The perceived benefit of this approach is release of the fluid while minimizing the risk of damaging normal ovarian tissue. Endometriomas treated this way have a high rate of recurrence (4). Multiple energy sources can be used to try and destroy the endometrioma wall. Electrical or radio-frequency energy (cautery), laser vaporization and plasma energy are common. Many promote these approaches as they feel they limit damage to normal ovarian tissue. However – with these approaches, there is no tissue to evaluate and no pathology to determine if normal ovarian tissue has been damaged or removed.
Excision of the endometriosis cyst – or cystectomy – is the process of actually removing the wall of the cyst (the functional part of the endometrioma). This is the part that produces the chocolate-like fluid. Some endometriomas are easily removed with blunt dissection or separation; others are more difficult and lack separation planes. Pathological evaluation of excised endometriomas can show some normal ovarian tissue. It is critical for the surgeon to understand dissection techniques and approaches to minimize the removal of normal cells.
—————————————————————–
As previously mentioned, we are concerned about the impact of surgery on ovarian reserve. AMH studies show an early postoperative decrease in AMH with a trend toward recovery 12 months afterward. If endometriomas are removed from both ovaries, there is a greater decrease in the AMH (5). The resulting “real-world” effect is that during egg retrieval for IVF, a fewer number of eggs may be retrieved for each cycle. It should also be noted, however, that simply by having severe endometriosis (6) and in particular endometriomas, AMH can be decreased (and may continue to decrease) whether surgery is performed or not. With proper disease removal and treatment of the endometrioma, AMH can and does improve in many cases.
The antral follicle count is determined by ultrasound and is the number of follicular cells that can be seen. The number either increases or remains unchanged after endometrioma excision.
It is interesting to note that the AMH and ANF give conflicting results with respect to fertility. One decreases and the other increases (7). It is important to remember that AMH and ANF are just lab or test markers. The ultimate goal of all of this fertility talk is successful pregnancy. Multiple studies have shown that successful pregnancy is more common after endometrioma removal than after simple drainage and cautery (8).
The technique used to remove the cyst wall is critical. Appropriate separation of the cyst, bleeding control and avoiding critical areas of the ovary are all important. Bleeding control or hemostasis is important, but still carries a risk of damaging normal ovarian tissue. Bleeding can be controlled by “cautery”, suturing or hemostatic agents that are used to help clot the small bleeding vessels. Cautery has been shown to decrease AMH levels more than the other two methods (9).
The first surgery for an endometrioma is the most important. The more surgeries that are performed for an endometrioma, the more damage that is done and the more difficult full/appropriate treatment becomes.
Aside from management of the endometriosis, we also concern ourselves with postoperative adhesions. When we perform radical excision of endometriosis whether or not it involves the ovaries, we always consider the risk of adhesions. Adhesion risk is greatest with higher levels of tissue damage or raw surfaces. Dense adhesions can form after surgery if two dissected areas or incisions come in contact. A full pelvic sidewall excision combined with removal of an ovarian endometrioma carries the risk of dense adhesion formation between the ovary and the side of the pelvis.
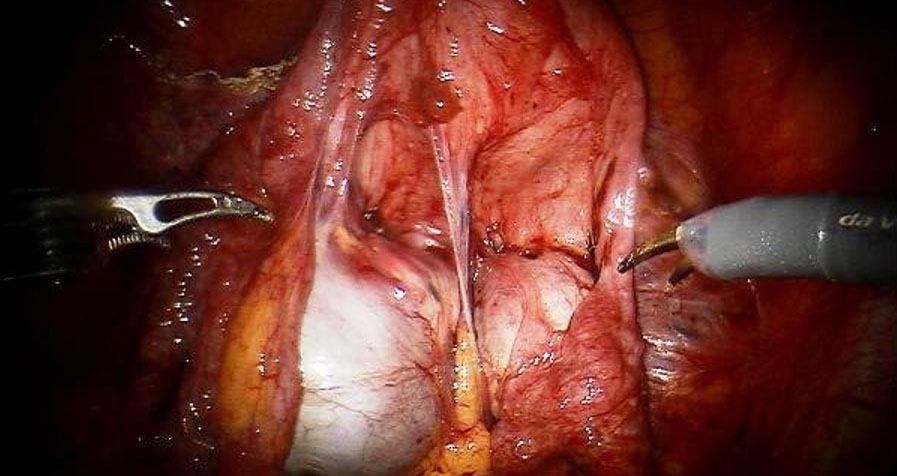
There are a few products that surgeons use off-label to minimize the risk of adhesion formations. Interceed, Seprafilm and amniotic membrane products are among those commonly used as adhesion barriers. Specifically, it is important to note that tight bleeding control is critical when using Interceed, as use in the presence of blood may increase adhesion formation. We also frequently use platelet rich plasma to coat the raw, surgical surfaces at the end of the surgery. This concentrates platelet rich growth factors which are the healing components of plasma.
As with other presentations of endometriosis, management of ovarian endometriosis and endometriomas must consider the patient as a whole. We cannot focus simply on fertility or simply on the cyst or simply on pain – we must consider each patient and how the endometriosis impacts each life. As physicians we share our knowledge with our patients to help each gain an understanding of the endometriosis and the options available. Each patient can then use this knowledge to make an informed, personal decision for care.
References1. Jenkins et al. Endometriosis: pathogenetic implications of the anatomic distribution. Obstet Gynecol. 1986; 67: 335–338.
2. Chapron et al. Ovarian endometrioma: severe pelvic pain is associated with deeply infiltrating endometriosis. Hum Reprod. 2012 Mar;27(3):702-11.
3. Goodman et al. Effect of surgery on ovarian reserve in women with endometriomas, endometriosis and controls. Am J Obstet Gynecol, 2016:Nov;215(5):589.
4. Vercellini et al. Surgery for endometriosis-associated infertility: a pragmatic approach. Hum Reprod. 2009 Feb;24(2):254-69.
5. Chang et al. Impact of laparoscopic cystectomy on ovarian reserve: serial changes of serum anti-Müllerian hormone levels. Fertil. Steril 2010 Jun;94(1):343-9.
6. Mostaejeran F, Hamoush Z, Rouholamin S. Evaluation of antimullerian hormone levels before and after laparoscopic management of endometriosis. Adv Biomed Res. 2015;4:182.
7. Muzii et al. Effect of surgery for endometrioma on ovarian function: a different point of view. JMIG. 2014, 21(4):531-3.
8. Hart et al. Excisional surgery versus ablative surgery for ovarian endometriomata. Cochrane Database Syst Rev. 2008, 16;(2):CD004992.
9. Ata et al. Effect of hemostatic method on ovarian reserve following laparoscopic endometrioma excision; comparison of suture, hemostatic sealant, and bipolar dessication. A systematic review and meta-analysis. JMIG. 2015: 22(3):363-72.
10. Seracchioli et al. The role of ovarian suspension in endometriosis surgery: a randomized controlled trial. JMIG. 2014, 21(6):1029-35.